 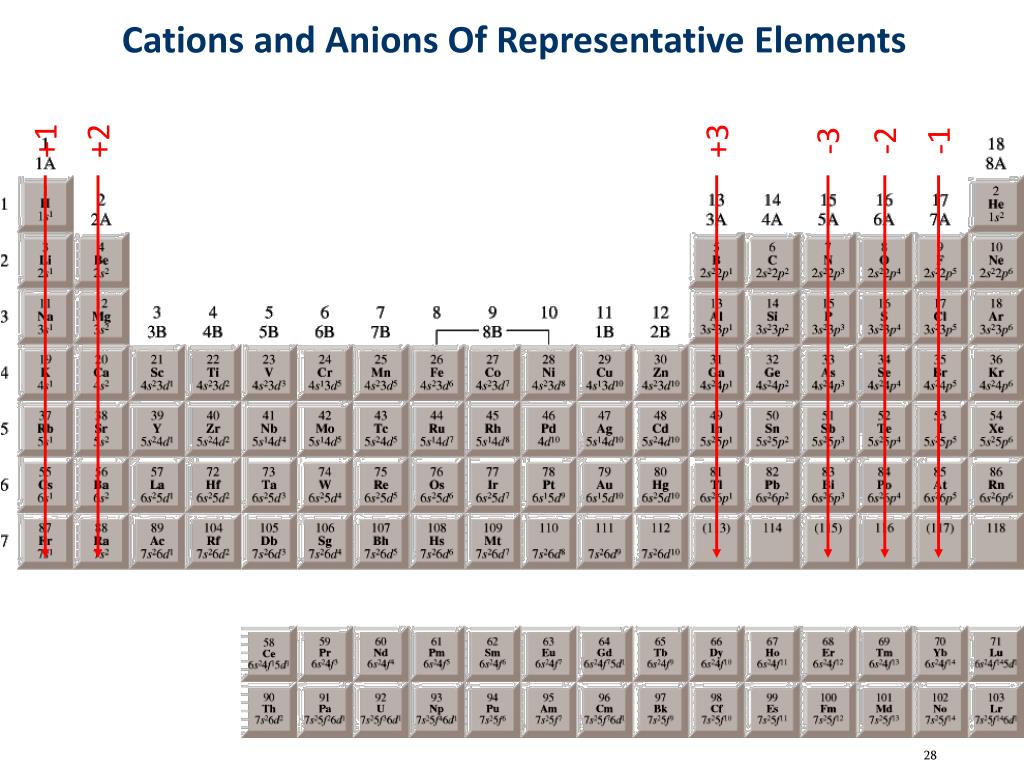
Anions are so named because they are attracted to the anode (positive field) while cations are attracted towards the cathode (negative field) in an electric field. For example, the fluoride ion is represented by the symbol F-, and the sodium ion is represented by the symbol Na+. The charge of an ion is indicated by a plus (+) or minus sign (-), which is written to the right of and just above the ion’s chemical symbol. Cation (metal) first Anion (nonmetal) second with -ide suffix The problem is some metals have more than one stable charge state, and so for those metals (type 2 metals), we need to identify the charge in the name. If m or n is 1, then no subscript is written in the formula. where the H 2 O is omitted if the is zero, m is the oxidation state of the anion, and n is the oxidation state of the anion. The formula of a salt is: (cation) m (anion) n ()H 2 O. 
For example, iron(II) has a 2+ charge iron(III) a 3+ charge. The number of valence electrons will be the key factor to determine if an element will be a cation or an anion. The rules are simple, name the cation first and the anion second, giving the anion the -ide suffix. Cations are always smaller than the neutral atom and anions are always larger. In other words, write the cation on the left and the anion on the right. Cations Ionic Charges Chart (Cations and Anions) Roman numeral notation indicates charge of ion when element commonly forms more than one ion. For example, when a sodium atom loses an electron, it becomes a positive sodium ion. Cation and Anion: Cations and anions are the established components of an ionic compound and it is held by the electrostatic interaction because of the opposite charges between the two ions. Positive ions, on the other hand, are just given the element name followed by the word ion. The charge of an electron is considered to be negative by convention and this charge is equal and opposite to the charge of a proton. An ion ( / a.n, - n /) 1 is an atom or molecule with a net electrical charge. Sizes of Common Cations and Anions of the Main Group Elements. Forming an ionic bond, Li and F become Li + and F ions. This identifies the element as manganese, Mn. Electron transfer between lithium (Li) and fluorine (F). Permanganate Ion MnO4- CID 24401 - structure, chemical names, physical and chemical properties, classification, patents, literature, biological activities, safety. The name is always the cation (usually metal) first then the anion + -ide ending. The substitution of inactive cation Ti for Fe and Mn is also beneficial to improve the electrochemical performance 80,81. o All elements alone have a charge of zero in their elemental state o. The anion F-doping in P2-Na 0.6 Mn 0.7 Ni 0.3 O 2. An easy mnemonic to distinguish the two is that the the word cation contains a plus sign (in other words, think of it as ca+ion). The use of one or more active or inactive elements to dope or replace Mn is beneficial to improve its performance. Like fluoride, other negative ions usually have names ending in –ide. An ion with a positive charge is called a cation, while an ion with a negative charge is called an anion. If atoms gain electrons, they become negative ions or anions. Examples of monatomic ions include Na +, Fe 3 +, Cl, and many, many others. This differentiates polyatomic ions from monatomic ions, which contain only one atom. The prefix poly- means many, so a polyatomic ion is an ion that contains more than one atom. If atoms lose electrons, they become positive ions or cations. In this article, we will discuss polyatomic ions. A few compounds of sodium, however, contain the Na ion, allowing comparison of its size with that of the far more familiar Na + ion, which is found in many compounds. Ions are atoms that have a positive or negative charge because they have unequal numbers of protons and electrons. Because most elements form either a cation or an anion but not both, there are few opportunities to compare the sizes of a cation and an anion derived from the same neutral atom.  
Here are some examples with determining oxidation states, naming a metal in an anion complex, and naming coordination compounds.Atoms cannot only gain extra electrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
